
Physical Activation Activated Carbon refers to a process that uses heat and gases to create a material with a very large surface area. You start by heating raw materials like coconut shells in a low-oxygen environment. Then, you treat the carbonized material with steam or carbon dioxide. This method helps remove pollutants from water, supports drug delivery, and boosts energy storage devices. You find it in industries that need to clean water, remove dyes, and improve batteries.
Key Takeaways
Physical activation uses heat and gases to create activated carbon with a large surface area, making it effective for water purification and air filtration.
Choose physical activation for cost-effective production and eco-friendly processes, while chemical activation is better for higher surface area needs.
Control the pore structure of activated carbon by adjusting temperature and the type of gas used during the activation process.
Physical activation is a cleaner method that avoids harsh chemicals, reducing waste and pollution, which supports environmental sustainability.
Common applications of physically activated carbon include water treatment, air filters, and gas storage, highlighting its versatility in various industries.
Physical Activation Activated Carbon vs. Chemical Activation
Main Differences
You can see clear differences between physical activation and chemical activation when you look at their process steps, materials, and outcomes. Physical Activation Activated Carbon uses heat and gases like steam or carbon dioxide to create pores. Chemical activation uses chemicals such as potassium hydroxide or phosphoric acid to help form pores at lower temperatures.
You might find it helpful to compare these methods side by side:
Activation Method | Cost Implications | |
|---|---|---|
Physical Activation | Low production costs, lower activation yield, less developed pore structure | Generally lower due to simpler processes |
Chemical Activation | Higher yields, more developed pore structures, requires acid washing | More costly due to additional processing |
Activation Method | Materials Used |
|---|---|
Physical Activation | Water vapor, carbon dioxide, oxygen, boron vapor, phosphorus |
Chemical Activation | Potassium hydroxide, potassium carbonate, sodium carbonate, magnesium chloride, phosphoric acid, sulfuric acid, aluminum chloride, sodium chloride, zinc chloride |
You get a well-developed pore structure and increased surface area with both methods, but chemical activation often produces higher surface areas and specific pore characteristics.
Type of Activation | Pore Structure Characteristics | |
|---|---|---|
Physically Activated Carbon | Well-developed pore structure, increased pore volume and surface area | CO2 adsorption capacity increases from 2.02 mmol/g to 2.37 mmol/g |
Chemically Activated Carbon | Higher surface areas and specific pore characteristics due to chemical activators | Total CO2 adsorption capacity increases from 1.07 mmol to 1.16 mmol |
When to Use Each Method
You should choose a method based on your needs and the application. Physical activation works well for industrial wastewater and air purification because it uses simple processes and eco-friendly materials. Chemical activation is better when you need activated carbon with a higher surface area or specific adsorption capacity.
Physical activation advantages:
Simple and cost-effective process.
Quick activation and ability to produce large quantities.
Uses abundant and inexpensive raw materials.
Chemical activation advantages:
Produces highly porous products at lower temperatures.
Offers more control over pore structure.
You can separate carbonization and activation steps in physical activation. In chemical activation, these steps happen together. If you want a process that is easy to control and uses fewer chemicals, physical activation is a good choice. If you need activated carbon with special properties, chemical activation may suit your needs better.
Physical Activation Activated Carbon Process

Carbonization Step
You start the process by heating raw materials like coconut shells, wood, or coal in a low-oxygen environment. This step is called carbonization or pyrolysis. The goal is to remove water and volatile substances from the material, leaving behind a carbon-rich structure. You usually heat the material to temperatures between 600°C and 900°C.
Typical temperature range for carbonization:
600–900°C
During carbonization, you create the first pores in the material. These pores are very small and are called micropores. The table below shows how the carbonization step helps form the initial pore structure:
Evidence Type | Description |
|---|---|
Micropore Development | Activation with CO2 leads to a micropore volume greater than 93%. |
Mesopore Formation | HNO3 activation forms some mesopores, but fewer than micropores. |
Pore Development Stages | The process opens inaccessible pores, creates new ones, widens existing pores, and merges pores by breaking walls. |
You can see that carbonization sets the stage for the next step by creating a network of tiny pores.
Activation with Steam or CO2
After carbonization, you move to the activation step. Here, you treat the carbonized material with oxidizing gases like steam or carbon dioxide. This step increases the number and size of pores, making the material much more effective for adsorption.
You usually use the following conditions for activation:
Initial pyrolysis temperature: 25°C to 500°C under nitrogen gas (flow rate: 500 cm³/min)
Activation with carbon dioxide gas (flow rate: 100 cm³/min)
Activation temperature range: 700–850°C
Holding time: 0–60 minutes
Activation temperatures can also range from 800°C to 1000°C when using steam or carbon dioxide.
The choice between steam and carbon dioxide affects the final properties of the activated carbon. The table below compares their effects:
Activation Agent | Effect on Porosity | Side-Produced Inhibitors | Functional Groups Formation |
|---|---|---|---|
CO2 | Enhances Microporosity | CO | Different functional groups |
Steam | Promotes Mesoporosity | H2 | Different functional groups |
CO2 helps you develop more micropores, which are good for adsorbing small molecules.
Steam creates more mesopores, which are better for larger molecules.
Temperature and Atmosphere
Temperature and the type of gas you use play a big role in how the pores develop. Higher temperatures help release more volatile substances and make the carbon more reactive. The atmosphere, or the mix of gases, also changes the way pores form.
Factor | Influence on Porosity Development |
|---|---|
Temperature | Higher temperatures promote the evolution of pore structures due to faster volatile release and higher reactivity. |
Atmosphere | The combination of oxygen and steam enhances pore structure formation through high reactivity and good diffusion. |
Activation Agents | Using CO2 and steam together leads to a synergistic effect, improving burn-off and porosity compared to individual use. |
You can adjust the temperature, gas flow, and activation time to control the size and amount of pores. This control lets you make Physical Activation Activated Carbon with the right properties for your needs. For example, you can create carbon with a very high surface area for water purification or with special pore sizes for gas storage.
Tip: If you want to boost both microporosity and mesoporosity, you can use a mix of steam and carbon dioxide during activation. This combination gives you a more versatile activated carbon.
By understanding each step and the effects of temperature and atmosphere, you can produce Physical Activation Activated Carbon that fits many industrial and environmental uses.
Science of Physical Activation
Formation of Porosity
When you use physical activation, you help create a network of tiny holes, or pores, inside the carbon. These pores form during both the carbonization and activation steps. The type of gas and the temperature you choose will change the size and amount of these pores. For example, carbon dioxide at high temperatures creates mostly micropores and ultra-micropores. Steam, also at high temperatures, gives you a wider range of pore sizes, including more mesopores.
Here is a table that shows how different activation agents affect pore structure:
Activation Agent | Temperature Range (°C) | Pore Structure Type |
|---|---|---|
Carbon Dioxide | High | Micro- and ultra-microporous |
Steam | High | Wider pore distribution, more mesopores |
You can also see how much space each type of pore takes up in the carbon:
Type of Pore | Porosity (%) | |
|---|---|---|
Micropores | 0.17 | 65.0 |
Mesopores | 0.71 | 65.0 |
Macropores | 3.3 | 79.7 |
High Macropores | 4.4 | 91.2 |
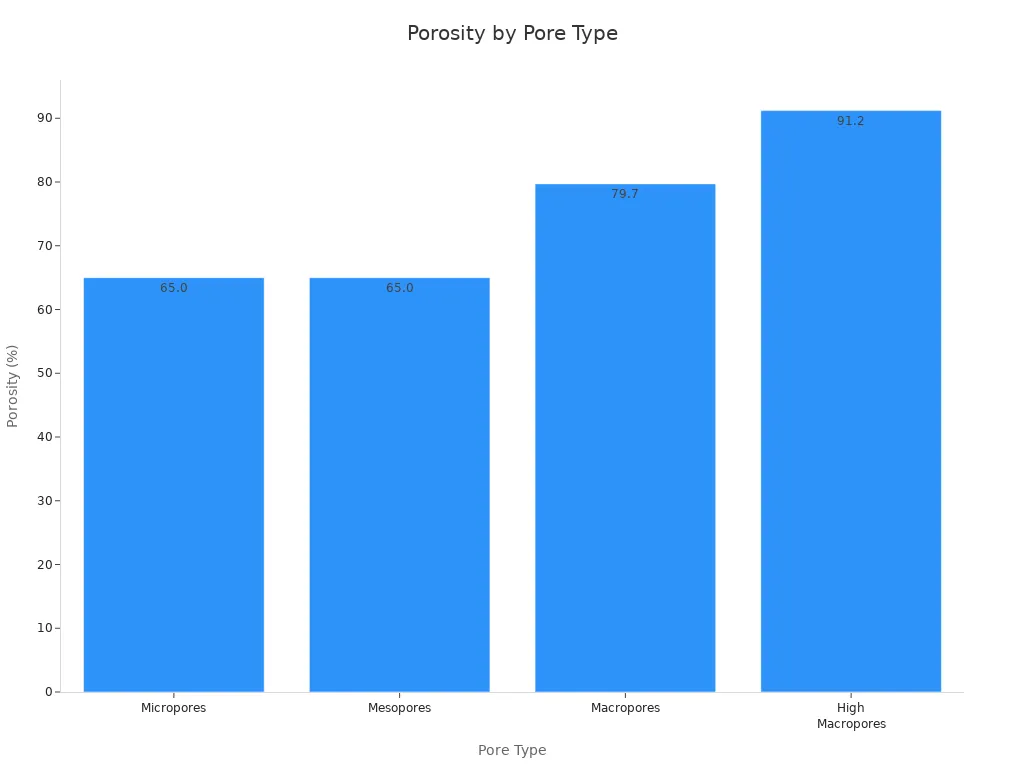
You can control the pore development by changing the activation method. For example, the two-step method uses carbonization at 400–600°C and then activation at 800–1100°C. This method increases the internal porosity by letting gases react with the carbon.
Surface Area Enhancement
Physical Activation Activated Carbon stands out because of its huge surface area. When you increase the number of micropores and narrow mesopores, you give the carbon more places to trap and hold molecules. This makes it very good for cleaning water, filtering air, or storing energy.
You can see how much the surface area grows after activation:
The surface area can go above 2000 m²/g.
Some methods give you even higher values, like 2540 m²/g after three hours of activation.
Conventional activation methods often reach about 2160 m²/g.
Tip: The more surface area you create, the better your activated carbon will work for adsorption tasks.
When you understand how porosity and surface area develop, you can make better choices for your projects. You can adjust the process to get the right balance of micropores and mesopores for your needs.
Industrial Methods and Applications

Common Equipment Used
You will find several types of equipment in factories that produce activated carbon. Each machine plays a special role in the process. Here is a table that shows the most common equipment and what each one does:
Equipment Type | Description |
|---|---|
Rotary Kilns | You use these for both carbonization and activation. They have a sealed drum that rotates inside a furnace. |
Fluidized Beds | You often use these for activation. They keep the carbon particles moving for even heating. |
Carbonization Kiln | You heat raw materials here to about 750°F to remove impurities and make pure carbon. |
Activation Kiln | You use this kiln at around 1,750°F. You inject steam to create activated carbon. |
Factories choose equipment based on the size of the operation and the type of raw material. Rotary kilns and fluidized beds help you control temperature and gas flow, which is important for making high-quality activated carbon.
Advantages and Limitations
You will notice many benefits when you use physical activation. Here are the main advantages:
The process is simple and cost-effective. You do not need to buy or handle dangerous chemicals.
You help the environment because you avoid chemical agents and do not create chemical waste.
You can control the pore structure and size, which improves the adsorption ability of the carbon.
The process is cleaner and does not need extra steps for wastewater treatment.
Physical Activation Activated Carbon is popular in water treatment, air purification, and gold recovery. You can use it to remove chlorine, odors, and harmful chemicals from water and air.
However, you should also know the limitations:
The process uses high temperatures, which can lower efficiency compared to chemical activation.
Some raw materials may not work well with physical activation.
You may need an acid wash after activation to remove ash or other residues.
Note: Many countries have rules for air pollution and waste. For example, the EPA in the USA and the EU’s Industrial Emissions Directive require factories to use clean and efficient methods. Physical activation helps you meet these standards because it does not create much pollution.
You can see that physical activation offers a balance of safety, cost, and environmental care. It works well for many industrial and environmental uses.
You can produce activated carbon through physical activation by following two main steps:
Heat raw material in an oxygen-free environment to remove volatile compounds.
Expose the carbonized material to steam or carbon dioxide at high temperatures to create pores.
This method gives you activated carbon with high thermal stability and efficiency for air purification, gas storage, and water filtration. When you select a production method, consider activation temperature, time, and the activating agent to optimize results.
Typical Uses | Practical Benefits |
|---|---|
Air filters, gas masks | High adsorption efficiency |
Metal extraction | Suited for high-temperature tasks |
Tip: Physical activation offers a cleaner, energy-intensive process that supports sustainability and future innovations.
FAQ
What raw materials can you use for physical activation?
You can use coconut shells, wood, coal, or nutshells. These materials have high carbon content and work well for making activated carbon.
Why do you need high temperatures in physical activation?
High temperatures help remove unwanted substances and open up pores in the carbon. This step makes the material better for adsorption.
Is physical activation safe for the environment?
Yes! You do not use harsh chemicals. The process creates less waste and pollution, making it a cleaner choice for the planet.
How do you know if activated carbon is good quality?
You can check the surface area and pore size. Good activated carbon has a large surface area and many tiny pores. You can also test its ability to adsorb substances.
Where do you use physically activated carbon?
Water purification
Air filters
Gold recovery
Gas storage
You find it in homes, factories, and even hospitals.
