
You use activated carbon in supercapacitor devices because it stores and releases energy quickly. When you choose materials for energy storage technology, you think about performance, cost, and sustainability. Most supercapacitors—about 70%—use activated carbon as the main electrode material.
You consider these factors when selecting materials:
Cost
Sustainability
Application requirements
Balance between energy and power
Stability and longevity
Environmental impact
Type | Power Density (kW/kg) | |
|---|---|---|
Traditional Batteries | 150 – 500 | N/A |
Supercapacitors | N/A | 10 – 20 |
Key Takeaways
Activated carbon is essential for supercapacitors due to its high surface area, allowing for efficient energy storage and quick release.
When selecting activated carbon, prioritize a good mix of pore sizes to enhance performance and ensure effective ion movement.
Using activated carbon from renewable sources, like coconut shells, is cost-effective and environmentally friendly, reducing waste.
Recent innovations in composite electrodes can significantly improve supercapacitor performance, offering higher energy storage and stability.
Stay informed about advancements in supercapacitor technology, as they promise to enhance energy storage solutions for various applications.
Activated Carbon in Supercapacitors

What Is Activated Carbon
You can think of activated carbon as a special form of carbon with a sponge-like structure. It has many tiny holes, called pores, that cover its surface. These pores give it a very high surface area, which means you can store a lot of charge in a small space. This property makes activated carbon very useful for energy storage devices like supercapacitors.
Activated carbon comes from different sources. You can make it from coconut shells, wood, or even waste materials like purple corncob. The way you produce activated carbon changes its properties. For example, you can use physical or chemical methods to create the pores. Chemical activation, especially with koh activation, is popular because it creates more pores and a higher surface area. You might also see other chemicals like H3PO4 or ZnCl2 used in the process.
Here is a table that shows how different activation methods affect the properties of activated carbon:
Activation Method | Surface Area (m²/g) | Specific Capacitance (F/g) | Capacitance Retention (%) | Notes |
|---|---|---|---|---|
KOH (two-step) | 4,048.2 | 419 | 91.2 after 20,000 cycles | High performance |
ZnCl2 | N/A | 201 | N/A | Moringa oleifera biomass |
KHCO3/KH2PO4 | N/A | 371.63 | Outstanding durability | Coconut shells |
N/S co-doped | N/A | 360 | 87 after 10,000 cycles | Lignosulfonate and polyaniline |
N-doped (squid ink) | N/A | 422.7 | N/A | Combined with ZnO |
KMnO4 | N/A | 333 | N/A | Fungus bran-derived |
KOH (reed straw) | N/A | 202.8 | 96.3 after 5,000 cycles | Nitrogen-rich carbon |
ZnCl2 (lotus stem) | 1,103.0-1,316.7 | 317.5 | 99.2 after 10,000 cycles | Balanced porous structure |
CMF-600 | 721.5 | 221 | N/A | Fibrous, mesoporous nature |
You can see that koh activation often leads to a very high surface area and strong performance. The choice of raw material and activation method changes how well the activated carbon works in a supercapacitor.
Why Activated Carbon Is Preferred
You might wonder why you should use activated carbon in a supercapacitor instead of other materials. The answer lies in its unique properties. Activated carbon has a high surface area and a network of pores. This structure lets it store more charge at the interface between the electrode and the electrolyte. When you use activated carbon, you get higher capacitance and faster energy storage.
The way you make activated carbon also matters. If you use koh activation, you can create more pores and increase the surface area. This helps the electrolyte move easily through the material, which improves the performance of the supercapacitor. You want the pores to be the right size. If they are too small, the electrolyte cannot get inside. If they are too big, you lose some of the benefits of the high surface area. The best activated carbon has a mix of tiny and medium-sized pores.
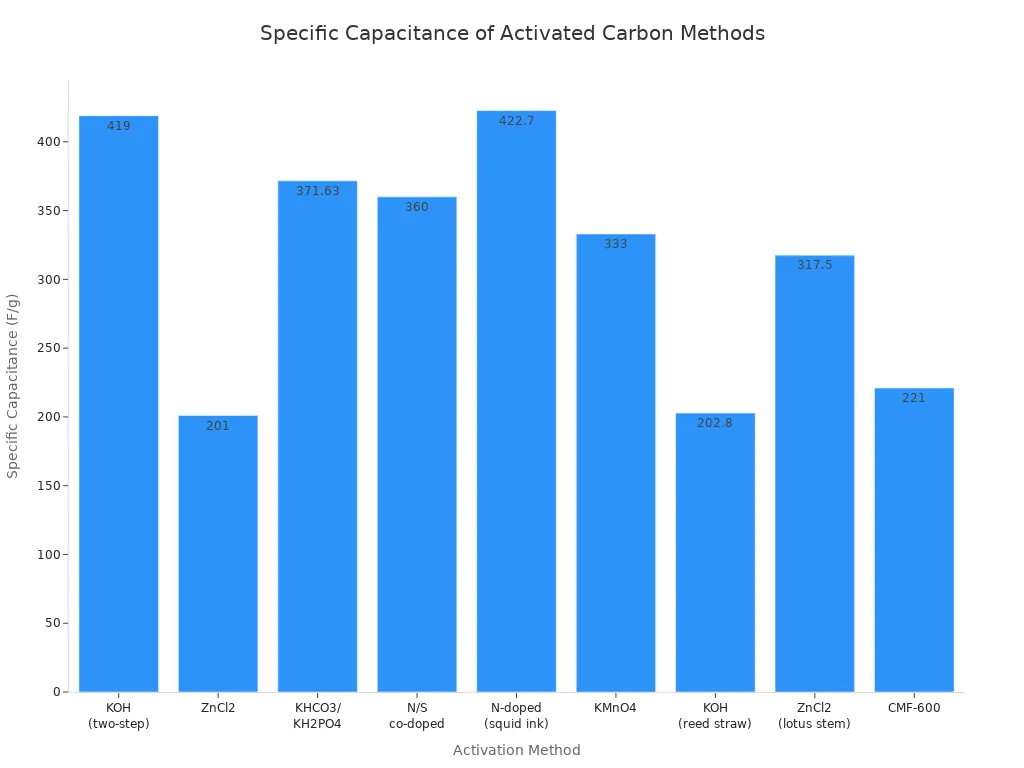
You can see from the chart that different activation methods give different results for specific capacitance. Koh activation stands out for its high values.
You also care about the source of the activated carbon. Many companies use waste biomass, like coconut shells or agricultural byproducts. This choice helps the environment and keeps costs low. You get a material that is both effective and sustainable.
Tip: When you choose activated carbon for a supercapacitor, look for a high surface area and a good mix of pore sizes. This will help you get the best performance.
Activated Carbon Electrodes

Structure and Function
When you build supercapacitor electrodes, you start with a thin layer of activated carbon. You place this layer on a metal sheet, often aluminum foil. This design helps the electrode conduct electricity well. The activated carbon has many tiny pores. These pores give the electrode a huge surface area. You can find values as high as 2,000 square meters per gram. This large surface area lets the electrode store more energy. The pores also help ions from the electrolyte move in and out quickly. This movement is important for fast charging and discharging.
You want the electrode to have both small and medium-sized pores. Small pores increase the surface area. Medium pores make it easier for ions to travel. When you use activated carbon electrodes, you get a balance between high energy storage and fast power delivery. This balance is why many people choose activated carbon for supercapacitor electrodes.
Deposition and Fabrication Methods
You have several ways to make activated carbon electrodes. One common method is to coat activated carbon onto aluminum foil. You can use a slurry of activated carbon and a binder. You spread this mixture on the foil and let it dry. This process creates a strong bond between the carbon and the metal.
Some advanced methods use special machines. For example, RF magnetron sputtering can deposit thin films on the electrode. In this process, you might:
Pre-sputter the surface at room temperature for a few minutes.
Use different times to control how thick the film becomes.
These steps help you get the right thickness and structure for your electrode.
You should remember that the way you make the electrode affects its performance. Chemical activation gives you more pores and a higher surface area. This means your supercapacitor electrodes can store more charge and last longer.
Tip: Always check the surface area and pore size when you choose activated carbon electrodes. These features decide how well your supercapacitor works.
Advantages of Activated Carbon
High Surface Area and Capacitance
You want your supercapacitor to store as much energy as possible. You can achieve this by using an electrode with a very high surface area. Activated carbon gives you this advantage. The surface of activated carbon is full of tiny pores. These pores create many places for charge to build up. When you use an electrode with more charge sites, you get higher capacitance.
The table below shows how surface area and pore structure help increase capacitance:
Evidence Description | Key Points |
|---|---|
High surface area of activated carbon | Increases charge accumulation sites on the electrode surface, leading to higher capacitance. |
Importance of pore structure | Open pores are crucial for effective ion transport and higher capacitance. |
Optimization of pore structure | Improves electrochemical characteristics and specific capacitance values. |
Role of ultramicropores | Boosts capacitance in AC-based electrodes. |
Porous carbon materials | Combine high electric conductivity and stability for higher specific capacitance. |
Specific surface area | Essential for achieving high capacitance. |
You can see that not all pores help with capacitance. Open pores let ions move in and out of the electrode quickly. This movement is important for fast charging and discharging. When you use activated carbon, you get a material that supports both high capacitance and quick energy flow.
Here are some measured capacitance values for supercapacitors using activated carbon electrodes:
Composition | Specific Capacitance (Fg−1) |
|---|---|
1:1 | |
1:2 | 266.3 |
2:1 | 250.2 |
These numbers show that you can reach high capacitance with the right electrode design.
Cost and Environmental Benefits
You care about cost and the environment when you choose materials for your electrode. Activated carbon stands out because you can make it from many sources, including waste biomass like coconut shells or corncobs. This makes it affordable and easy to find. You do not need rare or expensive materials.
When you compare activated carbon to other electrode materials, you see more benefits. Some advanced materials, like graphene or metal oxides, can give high capacitance, but they often cost much more and need complex processing. Activated carbon gives you a good balance between performance and price.
You also help the planet when you use activated carbon from renewable sources. You reduce waste and lower the carbon footprint of your device. This makes activated carbon a smart choice for both your wallet and the environment.
Tip: If you want a supercapacitor that is powerful, affordable, and green, choose activated carbon for your electrode.
Challenges and Innovations in Supercapacitors
Limitations of Activated Carbon
You may notice some challenges when you use activated carbon as an electrode in supercapacitors. One big issue is low conductivity. This means the electrode does not let electricity flow as easily as you want. Sometimes, activated carbon from biomass has tiny impurities. These impurities can cause unwanted reactions. They make the electrode less stable and reduce its electrochemical stability. You may also find that not all the pores in activated carbon are easy for ions to reach. This problem limits how much energy the electrode can store. These issues can lower the overall performance of your supercapacitor.
Recent Advances in Composite Electrodes
You can improve your supercapacitor by using new types of electrode materials. Scientists have made progress with composite electrodes. For example, they combine graphene with BiFeO3-BaTiO3. This mix helps the electrode store more energy and improves recycling. It also lowers costs. You may see other new materials, like graphene quantum dots and Cu-doped BiFeO films. These materials boost the performance of the electrode. They help you get higher energy storage and better electrochemical stability. Hybrid structures are also popular. When you mix activated carbon with graphene, you can get 30-50% better results than with activated carbon alone. If you add metal oxides or hydroxides, you increase energy density through fast redox reactions. Nanostructured designs, such as nanowires and 3D shapes, give the electrode more surface area. This makes it easier for ions to move and improves high power density.
Future Trends
You will see more changes in supercapacitor technology soon. Many researchers focus on hybrid systems. These combine the best parts of capacitors and batteries. The market for these hybrid systems is growing fast. It is worth about $1.2 billion now and could grow by 20% each year until 2028. The global supercapacitor market reached $3.27 billion in 2022 and may grow by over 15% each year through 2030. You will find supercapacitors in cars, renewable energy, and factories that need high power density. New designs make supercapacitors flexible and light. Some can do more than one job at a time. You will also see new electrode materials that focus on increasing energy density and stability. These trends show that supercapacitors will keep getting better and more useful for many applications.
You see that activated carbon plays a key role in supercapacitors. It gives you high surface area, tunable porosity, and strong stability. You can check the table below for a quick summary:
Benefit | Description |
|---|---|
High Surface Area | Stores more charge for better performance |
Cost-Effectiveness | Keeps devices affordable and scalable |
Environmental Advantage |
Recent research brings new composites and better pore design. You will find even more improvements as scientists use biomass and advanced structures. Stay curious about new energy storage materials—they shape the future of clean technology.
FAQ
What makes activated carbon special for supercapacitors?
You get a huge surface area with activated carbon. This lets you store more energy. The pores in the material help ions move quickly. You see fast charging and discharging.
Can you make activated carbon from waste materials?
Yes, you can use coconut shells, corncobs, or wood. Using waste helps the environment. It also lowers the cost of making supercapacitors.
How does activated carbon compare to graphene?
Activated carbon costs less than graphene. You find it easier to make. Graphene gives higher performance, but you pay more. For most uses, activated carbon gives a good balance.
Is activated carbon safe for the environment?
You help the planet when you use activated carbon from plants or waste. The process uses renewable resources. You reduce pollution and waste.
How long do activated carbon supercapacitors last?
You can expect thousands of charge and discharge cycles. Many devices last over 10,000 cycles. This means you get a long-lasting energy storage solution.
