
You see amazing energy storage every day, thanks to Super Capacitor Activated Carbon. This special material forms the heart of almost every commercial supercapacitor because it stores energy quickly and safely. Its unique structure gives you high energy density and reliable performance, making it the top choice over other materials like MXene or graphene. With Super Capacitor Activated Carbon, you get energy that charges fast and lasts longer—almost like magic.
Key Takeaways
Super Capacitor Activated Carbon stores energy quickly and safely, making it ideal for supercapacitors.
The unique pore structure of activated carbon allows for high energy density and fast charging, enhancing performance.
Activated carbon is cost-effective and easy to produce, making it the preferred choice over materials like MXene and graphene.
Supercapacitors using activated carbon can last over 10,000 charge cycles, providing long-lasting energy storage.
Applications of activated carbon supercapacitors include renewable energy storage, electric vehicles, and emergency power backup.
Super Capacitor Activated Carbon Basics

What Is Activated Carbon?
You might wonder what makes Super Capacitor Activated Carbon so special. Activated carbon is a form of carbon that has been processed to have a huge number of tiny holes, or pores. These pores give it a very high surface area, which means more space to store energy. You can think of it like a sponge, but instead of soaking up water, it holds electric charge.
The structure of activated carbon is unique. It contains three main types of pores: micropores, mesopores, and macropores. Each type helps store and move energy in different ways. Here is a table that shows the typical surface area and pore sizes you find in activated carbon for supercapacitors:
Parameter | Value Range |
|---|---|
1000–3000 m²/g | |
Micropore Size | 0.7–2 nm |
Mesopore Size | 2–50 nm |
Macropore Size | >50 nm |
Key Properties for Supercapacitors
You need activated carbon with the right properties to make supercapacitors work well. High surface area and the right mix of pore sizes help store more energy and move ions quickly. Production methods like calcination, pickling, and activation with chemicals such as KOH or potassium nitrate can change the pore structure and surface area.
Here are some common production methods and what they do:
Production Method | Description |
|---|---|
Two-step acid pickling | Removes impurities and introduces new porosity, improving activation efficiency. |
Direct activation | Uses KOH to create high surface area and excellent performance. |
Hydrothermal carbonization | Produces carbon with many types of pores and stable performance. |
A safer, single-step process that forms micropores. |
You get better energy storage when you use these methods to control the structure of Super Capacitor Activated Carbon. The right structure lets ions move in and out quickly, which means your supercapacitor can charge and discharge fast. This is why Super Capacitor Activated Carbon is so important for energy storage devices.
Why Activated Carbon Is Used
Surface Area and Porosity Advantages
You want your supercapacitor to store as much energy as possible. Super Capacitor Activated Carbon helps you do this because it has a very high surface area. This means there is more space for ions to gather and store charge. When you compare it to other materials, you see why it stands out:
Activated carbon gives you a surface area between 1000 and 2000 square meters per gram. This is much higher than many other materials.
The large surface area lets more ions stick to the surface, which increases how much energy you can store.
Materials like MXene and graphene can also store a lot of energy, but they have problems. For example, graphene layers can stick together, which lowers the surface area you can actually use.
You also need the right pore sizes in your activated carbon. Pores are tiny holes where ions move in and out. The mix of different pore sizes helps your supercapacitor work better. Here is a table that shows how each pore type helps:
Pore Size Type | Size Range (nm) | Functionality |
|---|---|---|
Micropores | 0.7–2 | Hold small ions and give you high surface area. If they are too small, ions move slowly. |
Mesopores | 2–50 | Let bigger ions move quickly. They help your device charge and discharge faster. |
Macropores | >50 | Act like highways for ions. They make it easier for ions to travel fast, especially when you need quick energy. |
When you use Super Capacitor Activated Carbon with the right mix of pores, you get high capacitance and fast charging. You can also improve the energy density and how long your device lasts by changing the pore structure during production.
Commercial Success and Cost Benefits
You want a material that works well and does not cost too much. Activated carbon is the only electrode material that you see in almost all commercial supercapacitors. It is popular because it is easy to make, and you can control its properties during production.
Let’s look at how the cost compares to other materials:
Material | Production Cost (per kg) | Scalability Issues |
|---|---|---|
Activated Carbon | $10-30 | Well-established manufacturing processes |
MXene | $200-1000 | Complex synthesis and limited industrial scale |
Graphene | $50-200 | Scalability challenges with high-quality production |
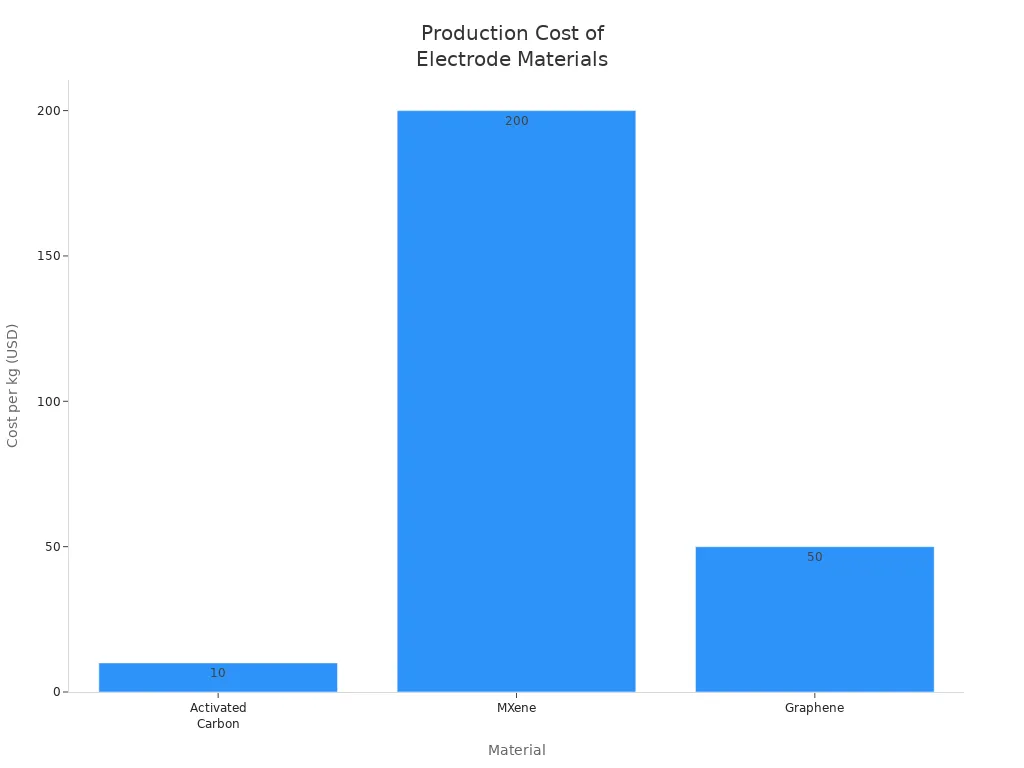
You can see that activated carbon costs much less than MXene or graphene. You also get a material that is easy to make in large amounts. This is why companies choose it for supercapacitors you use every day. You get reliable performance without paying a high price.
Tip: When you pick a supercapacitor for your project, look for ones that use activated carbon. You get the best mix of cost, performance, and long life.
How Activated Carbon Works in Supercapacitors

Charge Storage Mechanism
You might wonder how a supercapacitor can store so much energy so quickly. The answer lies in the way Super Capacitor Activated Carbon works at the molecular level. When you charge a supercapacitor, ions from the electrolyte move toward the surface of the activated carbon electrode. The high surface area of activated carbon—sometimes reaching up to 2000 square meters per gram—gives these ions plenty of space to gather.
Here is how this process helps you:
You get more energy storage because the large surface area allows more ions to stick to the electrode.
The porous structure of activated carbon lets ions move in and out very fast, so your device can charge and discharge quickly.
The right mix of micropores and mesopores helps ions travel with less resistance, which means you get higher power and better performance.
When the pore size matches the size of the ions in the electrolyte, you see even better energy storage and faster movement of charge.
You can see these features in the table below:
Parameter | Description |
|---|---|
Surface Area (1000–2000 m²/g) | More active sites for ion adsorption, which boosts capacitance and energy storage. |
Pore Size Distribution | Micropores store charge, while mesopores help ions move quickly for fast charging and discharging. |
Heat Treatment (800–1000°C) | Improves conductivity and keeps the structure strong, which is important for a good porous network. |
When you use Super Capacitor Activated Carbon, you get a material that stores charge by building up ions on its surface. This is different from batteries, which use chemical reactions to store energy. Supercapacitors use a physical process, so you get faster charging and longer life.
Electric Double-Layer Formation
The magic of energy storage in supercapacitors comes from something called the electric double layer. When you apply a voltage, positive and negative ions in the electrolyte move to the surface of the activated carbon electrode. They line up on the surface, forming two layers—one of ions from the electrolyte and one of electrons in the carbon. This is called the electric double layer.
The pore size of activated carbon must be close to the size of the ions in the electrolyte. A high surface area and a narrow range of pore sizes help ions move and build up charge. If the pores are too small, ions cannot move easily, which increases resistance and lowers performance, especially with certain electrolytes that allow higher voltages.
The resistance you see in a supercapacitor comes from the way ions move and form these double layers. If ions cannot move quickly through the pores, the device will not charge or discharge as fast.
You can compare this process to other ways of storing energy. Supercapacitors use the electric double-layer mechanism, which means they store energy by holding ions on the surface. Other materials, like those in pseudocapacitors, use chemical reactions to store more energy but may not last as long or charge as quickly.
Here is a table that shows the difference:
Mechanism Type | Pseudocapacitors | |
|---|---|---|
Charge Storage Mechanism | Electrostatic charge accumulation | Rapid and reversible Faradaic redox reactions |
Specific Capacitance | Lower | Higher |
Energy Density | Lower | Higher |
Charge Transport Process | Non-Faradaic (electrostatic) | Faradaic (electron transfer) |
Ion Interaction | Adsorption at the interface | Ion adsorption/insertion with redox reactions |
EDLCs, like those using activated carbon, store energy by building up charge at the interface between the electrode and the electrolyte.
Pseudocapacitors use fast chemical reactions, which can store more energy but may not offer the same speed or cycle life.
You can see why Super Capacitor Activated Carbon is so popular. It gives you fast charging, long life, and reliable performance. The electric double-layer mechanism makes it possible for you to use supercapacitors in everything from buses to backup power systems.
Performance and Comparisons
Benefits Over Other Materials
You get many advantages when you use activated carbon in supercapacitors. The high surface area gives you more places for ions to stick, which boosts energy storage. The pore structure helps ions move fast, so your device charges quickly. You also benefit from high purity, which keeps your supercapacitor stable and lasting longer. Activated carbon comes from renewable sources, making it eco-friendly and cost-effective.
Here is a table that shows how activated carbon stands out:
Performance Metric | Description |
|---|---|
Surface Area (1000–2000 m²/g) | More active sites for ion adsorption, enhancing capacitance and energy storage. |
Pore Size Distribution | Micropores store charge, mesopores allow faster ion movement. |
Purity (>99%) | Prevents unwanted reactions, improves stability and cycle life. |
Fast Charge/Discharge Capability | Allows rapid cycling without degradation. |
Eco-Friendly and Sustainable | Sourced from renewable materials. |
Cost-Effective and Scalable | Balances performance and low production cost. |
You see that activated carbon gives you a mix of high performance and low cost. It also supports fast charging, which is important for many modern devices.
Limitations and Trade-Offs
Activated carbon does have some drawbacks. Sometimes, large ions block the pores, which lowers performance. You may notice higher resistance in some supercapacitors. The energy density is not as high as in batteries or some advanced materials. Production can be complex and costly, especially if you want premium performance.
Pore blocking can occur with large ions.
Performance drops in non-aqueous electrolytes.
Higher resistance may limit power output.
Energy density is slightly reduced.
Production costs rise with advanced activation methods.
Activated carbon can degrade in humid or hot environments.
Note: Premium activated carbon costs more but gives you better performance. Lower-cost options may not last as long or work as well.
Real-World Applications
You see activated carbon supercapacitors in many places. They help stabilize power from solar and wind energy. Electric vehicles use them to recover energy during braking. Hospitals and data centers rely on them for emergency backup power. Portable devices charge faster because of supercapacitors. Rail systems and trams use them to save energy and cut emissions.
Renewable Energy Storage
Electric Vehicle Regeneration
Emergency Power Backup
Consumer Electronics
Transportation and Infrastructure
Supercapacitors with activated carbon help industries meet sustainability goals. They deliver quick bursts of energy and last through many charge cycles, making them valuable in many sectors.
You see how activated carbon gives supercapacitors their unique power. Heat treatment at 800°C boosts performance and improves conductivity, even with less surface area. The table below shows key findings:
Key Finding | Description |
|---|---|
Heat Treatment | Boosts electrochemical performance at 800°C. |
Conductivity | Improves even with reduced surface area. |
Durability | Maintains capacitance for over 10,000 cycles. |
Scalability | Biomass-based carbon works for large-scale use. |
You will find supercapacitors in many future technologies:
Electric vehicle regeneration
Emergency power backup
Consumer electronics
Transportation systems
You can appreciate how science shapes the devices you use every day.
FAQ
What makes activated carbon different from regular carbon?
You get activated carbon by heating regular carbon and adding steam or chemicals. This process creates millions of tiny pores. These pores give activated carbon a much larger surface area than regular carbon.
Can you recycle supercapacitors with activated carbon?
Yes, you can recycle supercapacitors. You can recover activated carbon and other materials. Recycling helps reduce waste and supports a cleaner environment.
How long does a supercapacitor with activated carbon last?
You can expect a supercapacitor to last for over 10,000 charge and discharge cycles. This is much longer than most batteries. You get reliable performance for many years.
Is activated carbon safe for the environment?
Activated carbon comes from renewable sources like coconut shells or wood. You can use it without harming the environment. It does not release harmful chemicals during use.
