
You can trust activated carbon to take out heavy metal pollutants from water very well. Science shows that metal removal activated carbon can remove up to 99% of many bad things in water.
SPO: 99.38%
CNS: 98.43%
MC: 99.40%
How much activated carbon you use, the pH of your water, and how much pollution is in the water all change the results.
Factor | Impact on Removal Efficiency |
|---|---|
Adsorbent Dosage | Using more gives better results by making more surface area. |
pH | The best results are usually between pH 3 and 9. |
These facts help you believe that activated carbon really works to clean water.
Key Takeaways
Activated carbon can take out almost all heavy metal pollution from water. This makes it a great way to clean water.
How well it removes metals depends on a few things. These include how much activated carbon you use, the pH of the water, and how much pollution is in the water.
Activated carbon works by trapping metal ions on its surface. It does this through physical and chemical adsorption.
Modified activated carbon can remove metals even better. Treatments that add special groups help it work more efficiently.
The size and porosity of activated carbon are very important. Smaller pieces with more pores have a bigger surface area. This helps them adsorb metals better.
There are different kinds of activated carbon. Some are made from wood, coal, or biomass. Each type has special features that change how well it removes metals.
It is important to check the water pH and metal levels often. This helps activated carbon work its best to remove heavy metals.
Activated carbon is used in many places. It helps clean water, treat industrial wastewater, and fix environmental problems. This keeps water cleaner and makes places safer.
Metal Removal Activated Carbon Mechanism
You may ask how activated carbon takes metals out of water. Scientists have looked at this closely. They found that activated carbon uses a few ways to trap heavy metals. The main ways are shown in the table below:
Mechanism Type | Description |
|---|---|
Physical Adsorption | Activated carbon has a very large surface area. This gives many spots for pollutants to stick. The pores are the right size for metal ions, so they get trapped. |
Chemical Adsorption | The surface of activated carbon has special groups. These groups, like phenolic hydroxyls, can grab heavy metal ions. This way is more picky and can still work after cleaning the carbon. |
Intermolecular Forces | Heavy metals are often ions. They stick to activated carbon with orientation forces. Organic pollutants use other forces called dispersion forces. |
Adsorption Process
Adsorption is the main way activated carbon removes metals. You can think of adsorption like a sponge soaking up water. But it happens on a much smaller scale. Activated carbon acts like a magnet for metal ions. It pulls them out of the water and holds them on its surface.
Physical vs. Chemical Adsorption
Physical adsorption happens when metal ions stick to the surface. This is because of weak forces. This process can be undone and is not very picky. You can wash the metals off the carbon. Chemical adsorption is different. Here, metal ions make stronger bonds with the surface groups. This way is more picky and hard to undo.
Physical adsorption can be undone and is less picky.
Chemical adsorption is more picky and hard to undo.
Things like temperature, pH, stirring time, and how much metal is in the water can change how well adsorption works. Higher numbers for these things usually mean better adsorption.
Scientists say it is important to know both the physical and chemical properties of activated carbons. This helps you pick the best one for your needs.
Role of Porosity
Porosity means how many tiny holes are inside activated carbon. These holes give it a huge surface area. More pores mean more places for metals to stick. Acid-activated carbons and biomass-derived activated carbon have special pore shapes. These shapes make them very good at removing heavy metals. The size and shape of the pores can match the size of metal ions. This helps improve how well adsorption works.
Ion Exchange
Ion exchange is another way activated carbon removes metals. In this process, metal ions in water swap with other ions on the carbon’s surface.
Studies show that used activated carbons can remove over 95% of lead and 86% of cadmium at low amounts. This is much better than new activated carbons, which only remove about 10-15%. Used carbons have more carboxyl groups on their surface. These groups help with ion exchange and make the carbon better at catching heavy metals.
Functional Groups
Functional groups are special parts on the surface of activated carbon. They help trap metal ions in different ways:
Electrostatic interaction
Ion exchange
Surface complexation
Physical adsorption
Precipitation
Groups with oxygen, like carboxyl and hydroxyl, help with ion exchange. Soft Lewis base groups, such as carbonyl, make dipole-dipole interactions stronger. Hard Lewis base groups, like deprotonated carboxylic acids, help with cation exchange. The pH of the water changes how these groups work. It affects the charge on the carbon and how well the groups can grab metal ions.
Carbon Modification
You can make activated carbon better at removing metals by changing its surface. Scientists use different ways to do this:
Adding magnetite, chitosan, or tannic acid can help remove chromium ions.
Chitosan makes activated carbon work better and last longer, especially at some pH levels.
Magnetite lets you pull out the carbon with a magnet, even if it lowers the surface area a bit.
Chemical treatments, like using acids or bases, add more functional groups to the surface.
Plasma treatment changes the surface without using liquids and can be used with other methods for better results.
These changes help activated carbon catch more metal ions. They also make it easier to use in water treatment.
Evidence for Heavy Metal Pollutants Removal
You can see that activated carbon works well for removing heavy metals by looking at what scientists found. They tested activated carbon in different ways. They used batch adsorption and adsorption–desorption tests to see how much metal comes out of water. These tests show why people trust activated carbon to clean up dangerous metals.
Research Findings
Lead, Mercury, Cadmium Removal
You might wonder how activated carbon removes lead, mercury, and cadmium. Scientists checked how much of each metal activated carbon can take out. Mercury is removed the most, especially with sulphurized carbon. Lead and cadmium are removed less, but pH changes do not affect them much. This means you get steady results for lead and cadmium, even if the water is more acidic or basic.
Contaminant | Notes | |
|---|---|---|
Mercury | Higher than Cadmium and Lead | Increased with sulphurized carbon; decreased at pH 2.0 |
Cadmium | Lower than Mercury | Less affected by pH changes |
Lead | Lower than Mercury | Less affected by pH changes |
You can remove more lead by using treated activated carbon. Treating the carbon adds special groups to its surface. These groups grab lead ions better. Studies show that treated activated carbon removes more lead than untreated carbon in both static and column tests.
Adsorbent Type | Method Used | |
|---|---|---|
Treated Activated Carbon | Higher efficiency | Static and column studies |
Untreated Activated Carbon | Lower efficiency | Static and column studies |
Scientists found that changing activated carbon with acids or other chemicals can double how much metal it catches. This means you get better results with modified activated carbons.
Copper, Chromium, Iron Removal
Activated carbon also works well for copper, chromium, and iron. Many studies use batch adsorption tests with different types of activated carbon. For example, activated carbon from banana, potato, orange, onion, opuntia, and pomegranate removes a lot of copper and chromium.
Metal | Source | |
|---|---|---|
Copper | 99.94, 100, 100, 100, 20.00, 90.00 | Banana, Potato, Orange, Onion, Opuntia, Pomegranate |
Chromium | 91.46, 100, 92.90, 93.40, 88.86, 100 | Banana, Potato, Orange, Onion, Opuntia, Pomegranate |
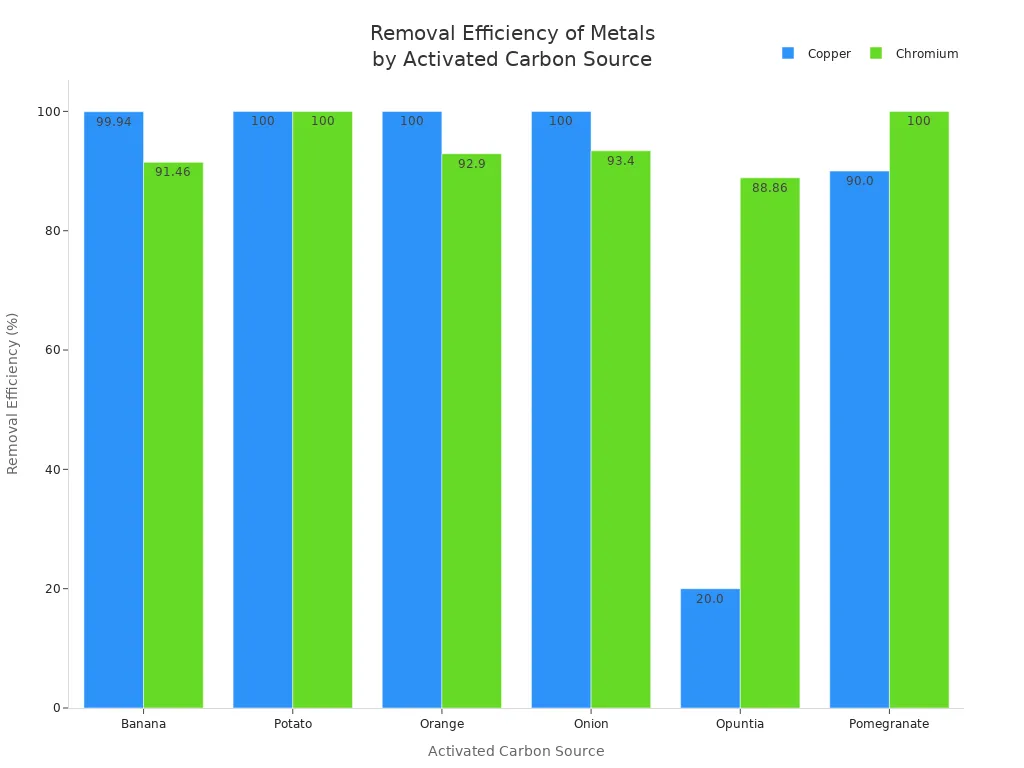
You can get even better copper removal by adding iron(III) to activated carbon. For chromium, the best removal happens when the water is more acidic. Low-cost activated carbon from African palm fruit works well for chromium, especially at low pH.
Removal Efficiency Data
Impact of Dosage
How much activated carbon you use changes how well it works. If you use a low dosage, you often see higher removal efficiency. This is because there are more metal ions for each spot on the carbon. When you use a high dosage, you still get good removal, but each gram of carbon takes up less metal.
Dosage Level | Removal Efficiency (%) | Metal Uptake (Co2+ ions) |
|---|---|---|
Low | Increased | Higher |
High | Increased | Decreased |
You can see this in studies with Conocarpus pruning waste. At low starting concentrations, copper and cadmium removal rates go above 85%. Even at higher concentrations, the rates stay above 50%. This shows that activated carbon keeps working well, even with more pollution.
Activated carbon from Conocarpus pruning waste removes over 85% of copper and cadmium at low concentrations.
At higher concentrations, removal rates stay above 50%.
This performance matches or beats other types of activated carbon.
Solution pH Effects
The pH of your water also changes how well activated carbon removes metals. Each metal has a best pH for removal. For example, chromium (Cr(VI)) is removed best at pH 3. Lead, copper, and zinc work best at pH 7. If you mix metals, zinc and cadmium removal can drop at pH 7.
Heavy Metal | Optimal pH | Adsorption Effect |
|---|---|---|
Cr(VI) | 3 | Optimal removal |
Pb(II) | 7 | Optimal removal |
Cu(II) | 7 | Optimal removal |
Zn(II) | 7 | Inhibited in mixed system |
Cd(II) | 7 | Inhibited in mixed system |
If you raise the pH from 3 to 5.9, lead and cadmium removal goes up.
Heavy Metal | Adsorption Effect | |
|---|---|---|
Pb(II) | 3 to 5.9 | Increased removal |
Cd(II) | 3 to 5.9 | Increased removal |
You should always check the pH of your water before using activated carbon. This helps you get the best results for heavy metal removal.
Factors Affecting Activated Carbon Performance
Carbon Type
You can pick from different activated carbon types for metal removal. Each type comes from a different source and has special features. Wood-based activated carbon is made from wood and plants. It has lots of pores and a big surface area. This helps it trap metal ions well. Coal-based activated carbon is made from coal. It is dense and works well in factories. Biomass activated carbon comes from coconut shells, fruit peels, or farm waste. It is good for the environment and works for many metals.
Wood-Based, Coal-Based, Biomass
Wood-based activated carbon removes metals like lead and copper well.
Coal-based activated carbon is best for tough water, like in factories.
Biomass activated carbon is eco-friendly and removes metals like chromium and iron.
You should choose the carbon type based on your water and which metals you want to remove.
Particle Size and Surface Area
The size of activated carbon grains and their surface area are important. Smaller grains give more surface area. This means more places for metal ions to stick. If you use tiny grains, metals come out faster.
The surface area of activated carbon is very important for adsorption. Bigger surface area means more spots for molecules to stick. This leads to better adsorption.
Activated carbon works well because of its shape, surface area, and pore sizes. Smaller grains make adsorption work better. The best results for bilirubin came from activated carbon with grains about 82 μm. The rate goes up when grains are smaller and mesopore volume is bigger.
Smaller grains and bigger mesopore volume make adsorption faster.
Activated carbon with small grains and big surface area removes metals better. You should look for these features to get the best results.
Water Chemistry
Water chemistry changes how well activated carbon works. You need to check the pH and metal concentration in your water. If pH is too low or high, activated carbon may not grab metal ions well. Metal concentration also affects how much gets removed.
pH and Concentration
You can see how pH and concentration change removal efficiency in the table below:
System Type | Metal Removed | Removal Efficiency | Influencing Factors |
|---|---|---|---|
Activated Carbon (AC) | Uranium (U) | Variable | pH, contact time, initial concentration, temperature |
Reverse Osmosis (RO) | Uranium (U) | >95% | pH, concentration polarization |
Reverse Osmosis (RO) | Manganese (Mn) | >95% | N/A |
Reverse Osmosis (RO) | Calcium (Ca), Manganese (Mn), Iron (Fe), Copper (Cu) | >98% | N/A |
You should set the pH to the best range for each metal. You also need to know how much metal is in your water. This helps you get the highest removal with activated carbon. If you use metal removal activated carbon, you can get better results by controlling these factors.
Dosage and Contact Time
You should think about how much activated carbon you use. You also need to think about how long it stays in the water. Both things change how well heavy metals are removed. Using more activated carbon gives a bigger surface area. This means there are more places for metal ions to stick. When you add more, you usually get better results.
Many studies show how dosage and contact time matter. Scientists saw that using more activated carbon helps remove more heavy metals. Letting the carbon stay in water longer also helps. More time gives metal ions more chances to stick to the surface.
Here are some key things to remember:
Using more activated carbon gives more places for metal ions.
Letting it sit longer helps more metal ions move to the carbon.
Higher temperature can help, but dosage and time are most important.
You can check the table below to see how these factors work together:
Factor | Effect on Metal Removal Efficiency |
|---|---|
Low Dosage | Fewer sites for metals, lower removal |
High Dosage | More sites for metals, higher removal |
Short Contact | Less time for adsorption, lower removal |
Long Contact | More time for adsorption, higher removal |
To get the best results, use enough activated carbon. Give it enough time to work. If you use too little, you might not remove all the metals. If you do not wait long enough, some metals may stay in the water.
This idea works for all types of activated carbon. It does not matter if it is wood-based, coal-based, or biomass. More carbon and more time always help.
Many water treatment plants use this idea. They add enough activated carbon and let water sit for the right time. This helps them take out dangerous metals and keep water safe.
If you use metal removal activated carbon at home or in a lab, always check the instructions. Make sure you use the right amount and let it work long enough. This easy step can help make your water much cleaner.
Limitations of Metal Removal Activated Carbon
You need to know that activated carbon does not remove all heavy metals the same way. Some metals are harder to get out of water. This can make things tricky when you use activated carbon.
Metals Difficult to Remove
Some heavy metals do not stick well to activated carbon. The table below shows how much each type can hold:
Heavy Metal | Activated Carbon Type | Adsorption Capacity (mg g-1) |
|---|---|---|
Cd (II) | Commercial AC | 0.7 |
Hg (II) | Commercial AC | 8.2 |
Cd (II) | Clothed AC | 3.8 |
Hg (II) | 145.0 | |
Cd (II) | Powder AC | 31.1 |
You can see that mercury sometimes sticks a lot, but cadmium does not. This means you might need special activated carbon or extra steps to remove these metals.
Arsenic, Chromium Challenges
It is even harder to remove arsenic and chromium. Scientists found some reasons for this:
People use cheap farm waste to make activated carbon because it costs less.
Researchers try to make bio-absorbents that need less heat, base, and acid.
Keeping the pH level steady is hard when removing heavy metals.
Activated carbon can be expensive, so you may not use enough.
You often need to change activated carbon to make it work better for arsenic and chromium.
It is tough to keep the best pH for removing these metals.
Saturation and Regeneration
Activated carbon cannot work forever. Over time, its pores fill up with metals and other stuff. When this happens, the carbon is called “saturated.” It will not work as well anymore.
When activated carbon is full, it cannot catch more metals.
You need to clean or replace it to keep your water safe.
Cleaning means treating the used carbon to get rid of the metals.
This lets you use the same activated carbon again.
If you do not clean it, you have to throw it away, which wastes money.
Operational Constraints
There are also some real-life problems when using activated carbon.
Cost and Maintenance
Activated carbon can cost a lot, especially if you need a big amount.
You may need to change or treat the carbon often to keep it working.
Making activated carbon better can also cost more money.
Disposal Issues
Used activated carbon with heavy metals is hard to throw away safely.
You must follow rules to get rid of it and protect nature.
If you do not do it right, you can pollute the environment.
Note: Scientists are still working to make activated carbon cheaper, safer, and better. New ideas may come soon, but you should always think about these problems before you pick a treatment method.
Applications of Activated Carbon

Water Treatment
Activated carbon is used in many water treatment systems. It helps take out heavy metals from drinking water. This keeps your water safe to drink. Scientists found that activated carbon can remove 75% to 96% of harmful metals. The removal rate depends on how much carbon you use. It also depends on the pH and which metals are in the water. Many cities use activated carbon in their water plants. This helps the water meet safety rules. You might see this if you visit a water treatment plant. The process protects your health by removing metals like lead, mercury, and cadmium.
Industrial Wastewater
Factories make wastewater with dangerous metals in it. Activated carbon can clean this water before it goes back to rivers or lakes. In these plants, activated carbon removes metals like mercury and lead. Coconut shell activated carbon can take out up to 80% of mercury and lead at low amounts. But it only removes about 29% of copper at the same amount. How much carbon you use and how much metal is in the water both matter.
Coconut shell activated carbon works best at about 600˚C.
Acid-activated coconut shell carbon removes more organic matter than other types.
Factories use activated carbon to follow environmental rules and keep water clean.
Activated carbon is important for making industrial wastewater treatment safer for everyone.
Household Filtration
You may use activated carbon at home and not know it. Many water filters in homes use activated carbon to remove metals and other bad things. These filters help take out hexavalent chromium and other harmful metals from tap water. Some systems mix activated carbon with reverse osmosis for better results. This gives you cleaner water for drinking and cooking. You can trust these filters to keep your family safe from dangerous metals.
Tip: Always read your filter’s instructions and change the activated carbon when needed. This helps your water filter work its best.
Activated carbon helps clean water in many places, from big factories to your kitchen. You can count on this technology to keep your water safe and clean.
Environmental Remediation
You can use activated carbon to help clean the environment. Scientists use it to take out heavy metals from soil, rivers, and lakes. When you use activated carbon for environmental remediation, you focus on places with pollution. These places can be near factories, mines, or old industrial sites.
Activated carbon traps heavy metals inside its pores. You can use it in many ways. Sometimes, you mix it into the soil. Other times, you add it to water or put it in special filters. This stops metals like lead, mercury, and cadmium from spreading. Using activated carbon helps protect plants, animals, and people.
Many engineers pick activated carbon because it works well and is easy to use. You can use it for water treatment and wastewater treatment. It helps in emergencies and in long-term cleanups. You might see it after chemical spills or in places with old pollution.
Here is a table that shows how well activated carbon removes different heavy metals during environmental remediation:
Heavy Metal | Success Rate (%) |
|---|---|
Lead | >90 |
Mercury | >95 |
Cadmium | up to 85 |
Chromium | 80-90 |
Arsenic | pH dependent |
Copper | 85-95 |
Zinc | 75-85 |
Nickel | 70-80 |
You can see that mercury and lead are removed the most. Cadmium, copper, and chromium are also removed well. Arsenic removal depends on the pH of the water or soil. You need to check the pH before you start your project.
Note: Activated carbon helps you meet strict safety rules for water and wastewater treatment. Cities and companies use it to make sure their cleanup projects work.
You might wonder what these projects look like. In one case, a team used activated carbon to clean a river near an old battery factory. They lowered lead and mercury by over 90%. In another project, workers mixed activated carbon into soil at a mining site. They stopped cadmium and chromium from getting into streams.
You can use activated carbon for many kinds of environmental remediation. It helps clean dirty water, treat bad soil, and keep your community safe. If you use it with other water or wastewater treatment methods, you get even better results. This makes activated carbon a smart choice for making the environment safer.
Innovations in Activated Carbon
There are many new ways to make activated carbon work better. Scientists are always trying to help us get cleaner water and a safer environment. These new ideas let us use activated carbon for more things, especially when cleaning up pollution.
Enhanced Carbon Materials
Now, there are special materials that make activated carbon stronger. These new materials can catch more heavy metals and help clean up the environment better.
Graphene oxide composites can hold much more lead and cadmium than normal activated carbon.
Some of these materials can grab 200 to 400 milligrams of metal for each gram you use.
Magnetic nanoparticles, like Fe3O4, let you pick up the activated carbon with a magnet. This makes cleaning up fast and easy.
You can use these magnetic materials again and again. They still work well after five times.
These new materials help you take out more pollution from water and soil. They give you better results when you clean up the environment.
Nanotechnology
Nanotechnology gives you more ways to clean up heavy metals. When you use nanotechnology, activated carbon works faster and better.
Carbon nanomaterials, like graphene and carbon nanotubes, have a huge surface area. This means more room for metals to stick.
You can change these nanomaterials with chemicals. This helps you catch certain metals when you clean up pollution.
Nanotechnology lets you clean water faster and with less waste.
You will see that nanotechnology makes activated carbon a good choice for many cleanup jobs.
Combined Treatment Methods
You can get even better results by using activated carbon with other ways to clean water. Many scientists mix activated carbon with things like filtration, ion exchange, or advanced oxidation.
You can use activated carbon with reverse osmosis to take out more metals from water.
Some people add special chemicals to activated carbon. This helps catch metals that are hard to remove, like arsenic or chromium.
You can use these mixed methods for big jobs, like cleaning rivers or treating factory water.
When you use these methods together, you make cleanup stronger and more dependable. You help keep your community and nature safe.
Tip: Always look for new ways to use activated carbon. These new ideas help you fix tough pollution problems and keep your water safe.
You can count on activated carbon to clean up heavy metals. Scientists say it works well for cleaning the environment. It removes a lot of metals from water and soil. Activated carbon has a big surface area. This helps it catch more metals. It is also a good deal for the price. The table below shows why it is a smart choice for cleaning up pollution:
Evidence Type | Description |
|---|---|
Effectiveness | Removal rates reach 75% to 96% in environmental remediation projects. |
Surface Area | Large surface area helps in environmental remediation tasks. |
Economic Viability | Safe, affordable, and sustainable for environmental remediation needs. |
When you use activated carbon, you help keep people and nature safe. You can expect new ways to make cleaning the environment even better.
